WHO Laboratory Biosecurity Guidance
In June 2024, WHO has published the revised Laboratory biosecurity guidance.
In June 2024, WHO has published the revised Laboratory biosecurity guidance.
As of May 2024, CWA 16335:2011 Biosafety professional competence standard has been converted to ISO/TS 5441:2024 Competence requirements for biorisk management advisors.
One of the ambitions of the Biosecurity Office is to provide information on biosecurity, including biosecurity and dual-use topics, to students in life sciences throughout their studies.

In the past few years, disinformation has frequently made headlines. Disinformation can be used as a weapon.

The Biosecurity Office regularly attends international meetings with the aim of sharing knowledge, acquiring knowledge, and networking.

There are regular reports being published that also address biosecurity.
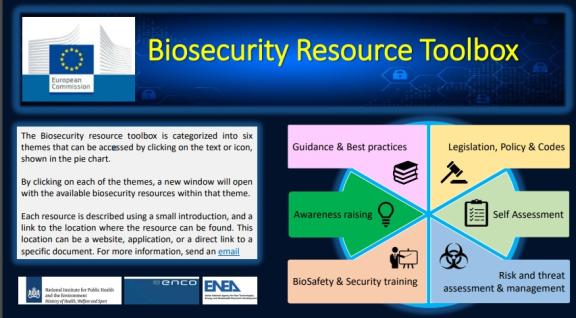
In the context of an EU project, Bureau Biosecurity developed a biosecurity resource toolbox together with consortium partners.
In the world of emerging technologies, such as synthetic biology, nanotechnology and nanobiotechnology, Safe-by-Design is becoming an increasingly common concept.

Instead of the Biosecurity Knowledge Day, Bureau Biosecurity organized a webinar on 19 November with the theme 'Biosecurity in Research'.