WHO Laboratory Biosecurity Guidance
In June 2024, WHO has published the revised Laboratory biosecurity guidance.
In June 2024, WHO has published the revised Laboratory biosecurity guidance.
As of May 2024, CWA 16335:2011 Biosafety professional competence standard has been converted to ISO/TS 5441:2024 Competence requirements for biorisk management advisors.
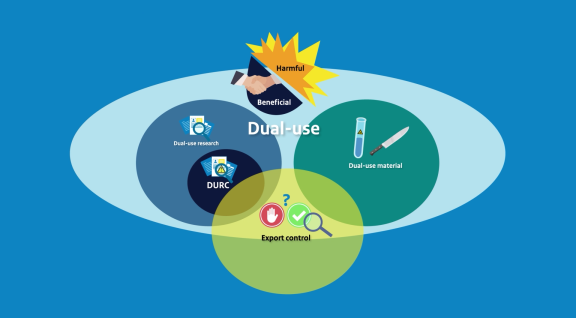
The Biosecurity Office has developed a new animation video about the concept of dual-use.
Pandemic preparedness, response, ethical hacking, export, a selection of the topics discussed during the 9th edition of the Biosecurity Knowledge Day.

The Biosecurity Office regularly attends meetings abroad with the aim of sharing knowledge, acquiring knowledge and networking.

Are laboratory employees sufficiently aware of how to act in the event of an unusual situation in the laboratory?
Artificial intelligence in combination with biotechnology offers unprecedented possibilities and rapid progress, but what are the dangers of AI?

The World Health Organization (WHO) published a report in October containing the findings of an international horizon scan on dual-use research of concern (DURC) in the life sciences.
It was exciting until the last minute whether it could go ahead, but we are happy that we can look back on a successful Knowledge Day.

In October, Rik Bleijs was elected co-chair of the steering committee of the International Experts Groups of Biosafety and Biosecurity Regulators (IEGBBR).
